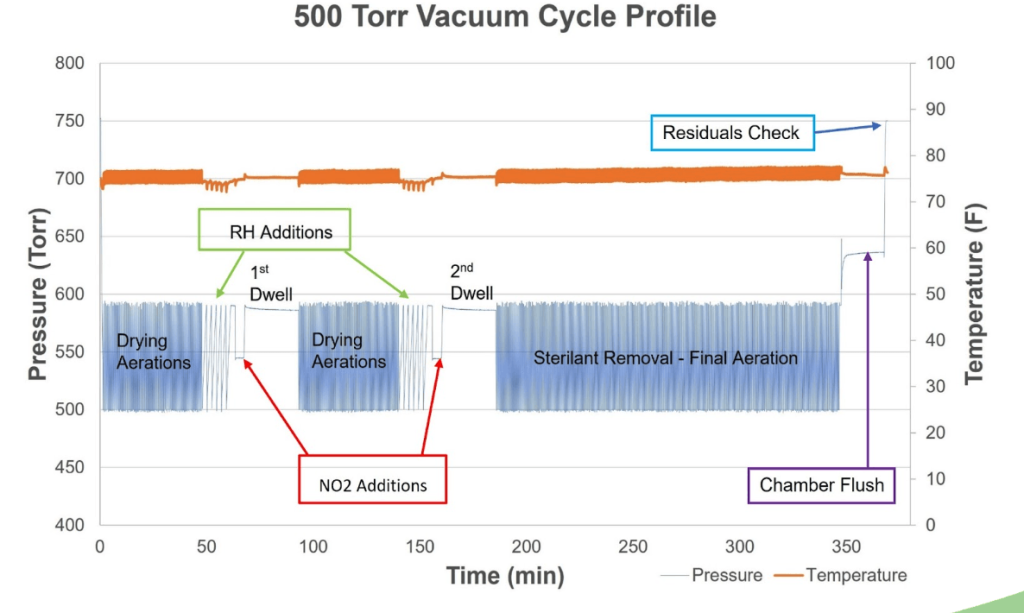
Applications
NO2 sterilization is the best choice for pre-filled syringes, combination products, and injectors
ADVANTAGES OF NO2 FOR PRE-FILLED SYRINGES:
-
Ultra-low temperature process
-
Minimum vacuum option prevents stopper movement
-
Low/no ingress through stoppers, maintains drug integrity
-
Flexible critical process parameters for custom cycles
-
Compatible with glass, polymer and lubricants
-
Safe and simple to bring in house, improves supply chain